Views: 0 Author: Site Editor Publish Time: 2026-02-26 Origin: Site











Tungsten Carbide is widely known for its strength and durability, yet many people still ask: does tungsten carbide rust? If you are considering it for jewelry or industrial use, understanding its corrosion resistance is essential. In this guide, we explain whether tungsten carbide can rust, oxidize, or corrode, and what environmental factors, material grades, and maintenance habits influence its long-term performance.
Before we answer does tungsten carbide rust, we need to understand what rust actually is. Many people use the word loosely. They call any surface discoloration “rust.” That is not accurate. Rust has a very specific meaning in chemistry.
Rust happens when iron reacts chemically in the presence of oxygen and water.
For rust to appear, three elements must exist:
Iron
Oxygen
Water
Remove one, and rust cannot form.
Rust only applies to iron or iron-based metals such as:
Carbon steel
Cast iron
Low-grade alloy steel
If a metal contains no iron, it cannot technically “rust.” It may corrode. It may oxidize. But it does not rust. This matters when people ask, does tungsten carbide rust? It contains no iron. So the classic rust reaction cannot occur.
People often mix these terms up. They sound similar. They are not identical.
Let’s break them down clearly.
| Term | What It Means | Applies To | Example |
|---|---|---|---|
| Rust | Iron forming iron oxide | Iron metals only | Steel turning reddish |
| Oxidation | Reaction between material and oxygen | Most metals | Aluminum forming Al₂O₃ |
| Corrosion | Gradual chemical breakdown | All materials | Acid damaging metal |
Here’s how to think about it:
Rust is a type of corrosion.
Oxidation is a chemical reaction involving oxygen.
Corrosion is the broader process of material damage.
For example:
Aluminum oxidizes.
Stainless steel resists rust.
Titanium forms a protective oxide layer.
We often see discoloration and call it rust. It feels easier. But scientifically, they are different. Oxidation can sometimes protect a surface. Aluminum does this. Titanium does it too. Rust behaves differently. It flakes. It spreads. It weakens the metal underneath. Corrosion may also happen without oxygen. Acids can cause it. Saltwater can accelerate it. Heat can speed it up.
When people ask does tungsten carbide rust, they usually picture reddish flakes forming on the surface. That kind of damage only happens to iron-based metals. Tungsten carbide contains tungsten and carbon, not iron. Because rust is specifically iron oxide (Fe₂O₃), the classic rust reaction simply cannot occur in this material.
Still, confusion happens. Many people use “rust” to describe any discoloration or surface change. In reality, rust is just one specific type of corrosion. If tungsten carbide changes appearance, something else is happening.
Tungsten carbide does not rust because it contains no iron. Rust requires iron, oxygen, and moisture reacting together. Remove iron from the equation, and the reaction stops before it begins.
Here’s a quick comparison:
| Material | Contains Iron | Can Form Rust |
|---|---|---|
| Carbon Steel | Yes | Yes |
| Stainless Steel | Yes | Sometimes |
| Tungsten Carbide | No | No |
So if you notice surface dullness or darkening, it is not rust. It may be oxidation, residue buildup, or environmental staining. The chemistry is completely different.
Yes, it can oxidize under high heat. Oxidation means a material reacts chemically with oxygen. It does not require iron.
For tungsten carbide, oxidation typically begins between 300–600°C (572–1112°F). At those temperatures, tungsten reacts with oxygen and forms tungsten trioxide (WO₃).
Temperature plays a major role:
Below 300°C → Stable in normal air
300–600°C → Slow surface oxidation may begin
Above 600°C → Oxidation becomes more noticeable
800°C+ → Rapid degradation in open air
In everyday environments, we never reach those levels. Jewelry, watch components, and household tools stay far below oxidation range. Industrial furnaces or cutting operations generate enough heat to trigger it.
So yes, it oxidizes under extreme heat. It does not rust at room temperature.
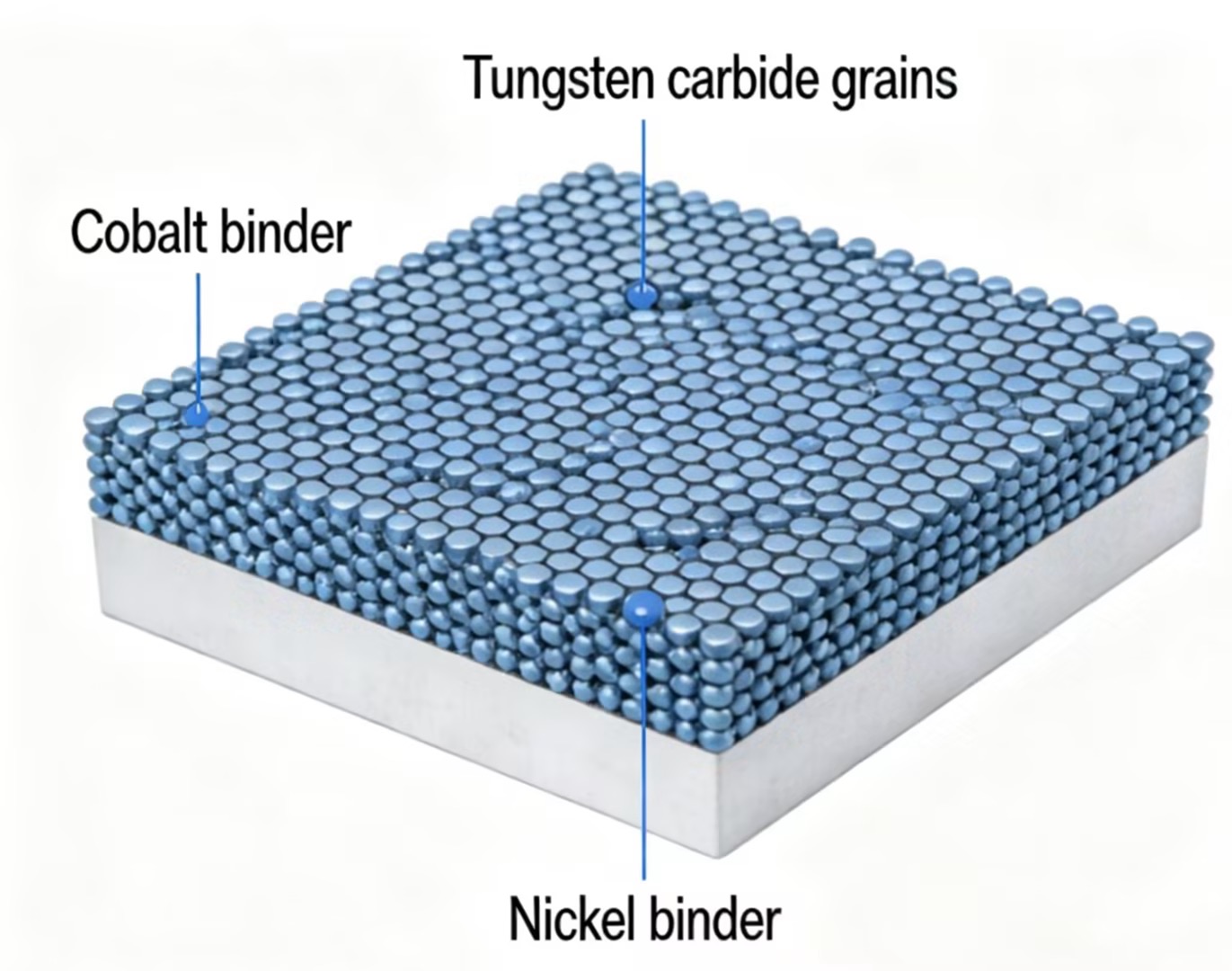
Corrosion is broader than rust. It refers to gradual material breakdown due to chemical reactions. Tungsten carbide itself is highly resistant, but most grades contain a binder metal. That binder holds the hard carbide grains together.
Common binders include:
Cobalt
Nickel
Cobalt-bonded grades are strong and widely used. However, cobalt can be vulnerable in aggressive environments. Strong acids, high salt exposure, and certain industrial chemicals may attack the binder phase first.
Here’s how the components compare:
| Component | Corrosion Resistance |
|---|---|
| Tungsten Carbide Grains | Very High |
| Cobalt Binder | Moderate |
| Nickel Binder | Higher than cobalt |
In harsh chemical settings, the binder may slowly degrade. Surface pitting can appear over long exposure periods. In normal daily use, this does not happen. Water, sweat, and air do not damage it.
When we ask does tungsten carbide rust, the practical answer for daily life is simple. It resists rust completely, handles moisture well, and remains stable unless exposed to extreme heat or aggressive chemicals.
We already know the answer to does tungsten carbide rust. It does not form iron rust. Still, its performance can change depending on environment and material quality. Corrosion resistance is not a simple yes-or-no issue. Several factors influence how it behaves over time.
The surrounding environment plays a major role. Even strong materials react differently under stress.
Humidity
Moist air alone does not cause rust in tungsten carbide. It contains no iron. However, long-term moisture exposure may slowly affect the metallic binder phase.
Salt Exposure
Salt increases electrical conductivity. It speeds up corrosion in many metals. Coastal areas. Marine equipment. Offshore platforms. They create harsher conditions.
Effects of salt-heavy environments:
Faster binder degradation
Possible surface pitting over long periods
No formation of iron oxide
Temperature
Heat changes everything. As temperature rises, chemical reactions accelerate.
| Temperature Range | Effect on Tungsten Carbide |
|---|---|
| Room temperature | Stable and corrosion resistant |
| 300–600°C | Surface oxidation may begin |
| Above 600°C | Faster oxidation possible |
| Extreme heat cycles | Structural stress risk |
Normal outdoor heat does not cause problems. Industrial furnaces create different conditions.
Chemicals often matter more than moisture.
Strong Oxidizers
Highly oxidizing acids can attack binder metals. Nitric acid. Concentrated oxidizing solutions. They break down metallic phases first.
Possible effects include:
Surface dulling
Localized corrosion spots
Reduced structural cohesion over time
Halogens (Chlorine, Fluorine)
Halogen elements are highly reactive. Chlorine in high concentration can degrade cobalt binders. Fluorine is even more aggressive.
Here’s a simplified comparison:
| Chemical Type | Risk Level |
|---|---|
| Neutral water | Very low |
| Mild alkaline solution | Low |
| Strong oxidizing acids | Moderate to high |
| Halogen-rich environment | High |
Under typical daily exposure, none of these cause issues. Industrial chemical plants require careful grade selection.
Not all tungsten carbide is identical. Composition changes performance.
Most industrial grades fall into two main categories:
WC-Co (Tungsten Carbide–Cobalt)
WC-Ni (Tungsten Carbide–Nickel)
Cobalt binders provide excellent toughness. They are common in cutting and drilling tools. However, they show moderate corrosion resistance in chemical environments. Nickel binders offer better corrosion resistance. They perform well in marine or chemical settings.
| Grade Type | Corrosion Resistance | Typical Use |
|---|---|---|
| WC-Co | Moderate | Mining, machining |
| WC-Ni | Higher | Chemical processing |
| Alloyed grades | Enhanced | Specialized applications |
Advanced alloyed versions may include chromium or other elements. These additions improve oxidation resistance and chemical stability. When we ask does tungsten carbide rust, the grade matters for corrosion behavior, even though rust itself never forms.
Production quality affects long-term durability.
Density
Higher density means fewer internal gaps. Fewer gaps reduce pathways for chemical penetration.
Porosity
Porous material allows fluids to enter micro-voids. That increases corrosion risk in aggressive environments.
Surface Finishing
A smooth, polished surface resists corrosion better than a rough one. Scratches create micro-sites for chemical attack.
Here’s how manufacturing factors influence performance:
| Factor | Impact on Corrosion Resistance |
|---|---|
| High density | Improves durability |
| Low porosity | Reduces chemical penetration |
| Fine surface finish | Minimizes localized attack |
| Poor sintering control | Increases vulnerability |
Two components may look identical. Their internal structure may differ significantly. Quality control makes a noticeable difference over years of service. When evaluating whether tungsten carbide rusts or corrodes, we need to consider environment, chemistry, composition, and manufacturing precision together.
Clean it using mild soap and warm water. A soft cloth works well. Rinse it thoroughly, then dry it completely using a lint-free towel. Proper drying prevents mineral spots and surface buildup. We should also inspect it regularly. Look for scratches, dull areas, or minor pitting in industrial parts. Small surface issues are easier to manage early.
Store it in a dry, stable environment. Normal room temperature is ideal. Avoid damp areas such as bathrooms or poorly ventilated workshops. Use protective packaging if possible. Soft pouches, sealed containers, or protective wrapping reduce dust and accidental scratches. Industrial components benefit from climate-controlled storage, especially in humid regions.
Avoid harsh chemicals such as strong acids or bleach-based cleaners. They may attack binder metals over time. Do not use abrasive scrubbers or polishing powders. They create micro-scratches on the surface. Limit prolonged chlorine exposure in high-concentration environments. While everyday contact causes no rust, aggressive chemical exposure can gradually reduce surface integrity.

A: No. Tungsten carbide does not rust because it has no iron. However, binder metals like cobalt may corrode in harsh chemical environments.
A: It rarely turns green. Discoloration may come from skin reactions or binder metals, not rust.
A: Yes. Water does not damage or rust it under normal conditions.
A: It may dull slightly over time due to surface scratches, not rust.
So, does tungsten carbide rust? No, it does not form traditional rust because it contains no iron. It offers excellent resistance to moisture and daily wear, though extreme heat or harsh chemicals can affect certain binder grades.
Choosing high-quality material makes a difference. At NJ-ModernDiamond Co., Ltd., we provide precision-engineered tungsten carbide solutions designed for durability, reliability, and long service life.
